  
Thomson, led to the discovery of the negatively charged part of the atom, the electron. Thomson also concluded that electrons are part of atoms. The cathode ray tube experiment, originally carried out by J.J. Thomson proposed the plum pudding model of the atom, which had negatively-charged electrons embedded within a positively-charged 'soup. In 1897 he showed that cathode rays (radiation emitted when a voltage is applied between two metal plates inside a glass tube filled with low-pressure gas) consist of particles electronsthat conduct electricity. Before his work, atom was considered to be. Thomson detected their path by the fluorescence on a squared screen in the jar. Thompson performed a brilliant experiment which proved that atom consisted of charged subatomic particle(s). Cathode rays were produced in the side tube on the left of the apparatus and passed through the anode into the main bell jar, where they were deflected by a magnet. This was the beginning of further understanding leading to the atomic theory and structure that we know today.Ĭathode ray tube is a tube that contains a small amount of gasīetween two metallic plates. Thomson's experiments with cathode ray tubes showed that all atoms contain tiny negatively charged subatomic particles or electrons. Thomson first investigated the magnetic deflection of cathode rays. He won the 1906 Nobel prize for the discovery of electrons. He zapped atoms with electricity and observed that negatively charged particles were removed! He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles. 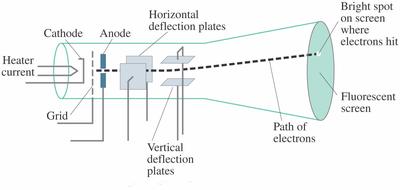
Thomson, a British physicist, conducted the cathode ray experiment. Thomson determined that charged particles much lighter than atoms, particles that we now call electrons made up cathode rays. A cathode-ray tube (CRT) is a vacuum tube in which an electron beam, deflected by applied electric or magnetic fields, produces a trace on a fluorescent screen. Previously, atoms were known to be indivisible, but in 1897, J. Thomson (1897) who discovered electron) extended the cathode ray experiment for the determination of velocity of electrons and their charge/mass. Thomson knew that the cathode ray tubes that they had only work if there is a little air in the tube and the amount of air needed depended on the shape of the.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
